Updated January 21, 2022
Due to the risk of Thrombosis with Thrombocytopenia Syndrome (TTS), a rare but serious adverse event, the CDC now recommends the use of mRNA vaccines (Pfizer or Moderna) over the Janssen/J&J vaccine for all vaccine-eligible people. Vaccine providers should start the two-dose mRNA vaccine series even if there is uncertainty about how the patient will receive their second dose; setting alone should not be a reason to offer the J&J vaccine. However, J&J may be offered in some situations as described below:
- When there is a contraindication to mRNA vaccines (e.g., severe allergic reaction after a previous dose or to a component of an mRNA vaccine)
- When a person would otherwise remain unvaccinated for COVID-19 due to limited access to vaccines.
- When a person wants to receive J&J despite the safety concerns identified
For more information, please review the CDC’s Interim Clinical Considerations webpage.
Previous updates:
Warning of rare Guillain-Barré syndrome cases, July 2021
On July 13, the FDA announced an update to the Johnson & Johnson (J&J) COVID-19 vaccine to include a warning about Guillain-Barré syndrome (GBS). GBS is a neurological disorder in which the body’s immune system damages nerve cells, causing muscle weakness, or in the most severe cases, paralysis. So far there have been 100 reports of GBS out of the 12.5 million doses administered, a rate of 1 in 125,000 so far. Of these reports, 95 of them were serious and required hospitalization. There was one reported death. These cases have mostly been reported about two weeks after vaccination and in males ages 50 and older. Although the available evidence suggests an association between the J & J vaccine and increased risk of GBS, it is insufficient to establish a causal relationship. No similar signal has been identified with the Moderna and Pfizer-BioNTech COVID-19 vaccines.
Actions requested of clinicians:
- Inquire about recent J & J COVID-19 vaccination in any patient presenting with signs or symptoms consistent with GBS (i.e. weakness or tingling sensations, especially in the legs or arms, that are worsening and spreading to other parts of the body; difficulty walking or with facial movements, including speaking, chewing or swallowing; double vision or inability to move the eyes; difficulty with bladder control or bowel function).
- Report cases of GBS after vaccination promptly to the U.S.Vaccine Adverse Events Reporting System (VAERS).
- Review the updated J&J EUA Fact Sheet for Providers and the J&J EUA Fact Sheet for Caregivers and Recipients.
Resumption of vaccinations and warning of rare blood clots, April 2021
On April 23, after a 10-day pause, the CDC voted to resume vaccinations with the Johnson & Johnson (J&J or Janssen) vaccine for people ages 18+ because the benefits far outweigh the risks of very rare blood clots. Meeting slides and data updates discussed at the April 23rd CDC Advisory Committee on Immunization Practices (ACIP) meeting can be downloaded here.
Read CDC questions and answers on the J&J vaccine here and the detailed CDC process, analysis and recommendations on the J&J vaccine here.
Click here for J&J updates from California state and fact sheets in multiple languages.
More background, data and clinical guidance:
The CDC and FDA paused J&J vaccinations on April 13 because there had been 6 cases of rare and serious blood clots reported called cerebral venous sinus thrombosis, or CVST, combined with thrombocytopenia, or low platelet counts, out of the nearly 7 million doses adminstered. The pause provided time for health providers to be alerted and identify and treat more cases and gather more information.
On April 23rd the CDC voted to resume vaccinations with the original approval for people ages 18 and over. This condition is now called the thrombosis with thrombocytopenia syndrome (TTS) following Janssen COVID-19 vaccine.
Dr. Sarah Oliver of the CDC presented a brilliant analysis of risks and benefits which showed that by resuming J&J vaccinations for all people ages 18+, while there might be 24-45 cases of this rare clotting syndrome, there would be 800 to 3,500 fewer ICU admissions due to COVID-19 and 600-1,400 fewer deaths due to COVID-19. Based on this analysis far more lives would be saved compared estimated number of cases of the rare clotting syndrome.
On May 12, Dr. Shimabukuro from the CDC reported that there were a total of 28 cases of TTS clots out of the over 9 million J&J vaccines given in the US, all in adults ages 18-59, with 22 cases in women and 6 in men. 3 people have died. For women ages 20-50 years, this is higher than the background rate for CVST.
Overall, the risk for the TTS clots remains extremely low at about 3 cases per 1 million. There have been 12 cases per million doses among women between the ages of 30-39 and 9 cases per million doses among women between 40 and 49, the two demographic groups that appear to be at highest risk.
Details can be found in this CDC health alert, the JAMA study on the first 12 J&J clot cases, slides with case and data updates from the April 23 CDC ACIP meeting, the CDC update on May 12, and in this April 30 CDC MMWR article summarizing the ACIP J&J vaccine recommendations.
On April 20, the European Medicines Agency announced that the risks of the clots are “very rare” and that the overall benefits of the J&J vaccine “outweigh the risks of side effects.”
There are no reports of these rare types of clots with low platelet counts in the over 220 million doses of the Moderna or Pfizer mRNA vaccines administered so far.
To put the risk of these clots in perspective:
- 3 in 1 million people have had clots and low platelets after the J&J vaccine
- 5-6 in 1 million hospitalized with COVID-19 have had the CVST clot
- 8-10 in 1 million have had clots with low platelets after the AstraZeneca vaccine
- 9-12 in 1 million women ages 30-49 have had clots and low platelets after the J&J vaccine
- 15-28 in 1 million develop the CVST clot each year
- 30 in 1 million have died from COVID-19 in the US
- 200 in 1 million have been hospitalized with COVID-19 in the US
- 1,000-2,000 in 1 million develop clots each year from air travel, birth control pills, injury and immobility (learn more about clots here)
What should we do about this?
People who have received the J&J vaccine in the past month:
- If you develop severe headache, abdominal pain, leg pain, or shortness of breath within a month after vaccination, contact your health care provider and seek care right away.
Health care providers and staff:
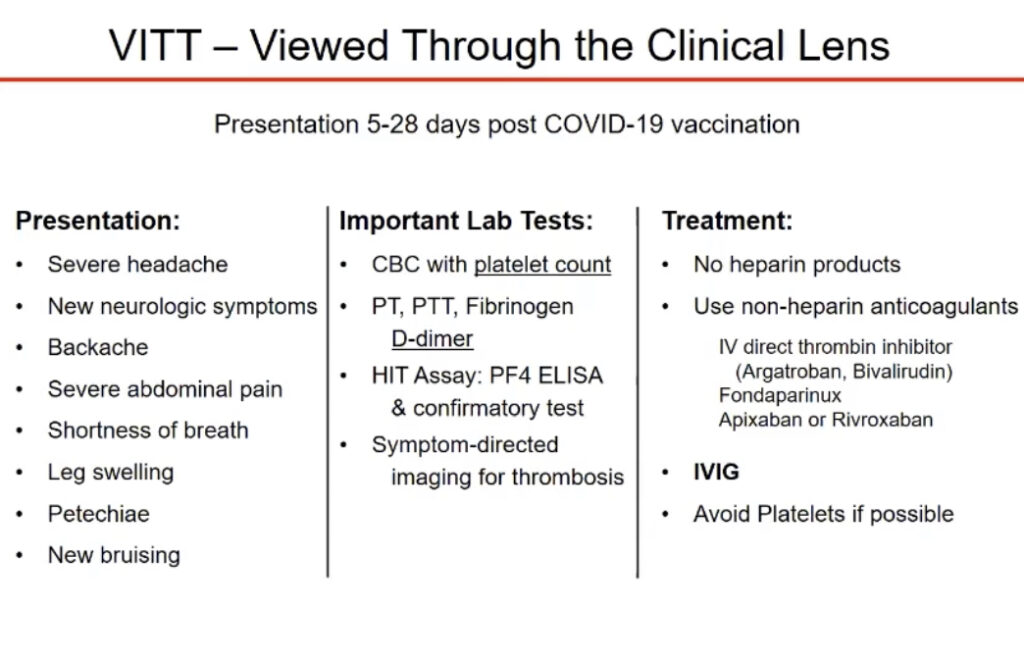
- Recognize symptoms of clots and low platelets: severe headache, backache, new neurologic symptoms, severe abdominal pain, shortness of breath, leg swelling, petechiae (tiny red spots on the skin), or new or easy bruising.
- Evaluate and treat: consult a hematologist; check platelet counts, PF4 ELISA (labs for HIT) and platelet activation assay; avoid heparin to treat clots with low platelet counts (<150k/ml) in people who’ve received the J&J vaccine. Instead consider non-heparin anticoagulants (such as argatroban) and high-dose intravenous immune globulin (IVIG). The American Society of Hematology (ASH) has released guidance for diagnosis and clinical management and the CDC presented Pathogenesis and Management of Thrombosis with Thrombocytopenia Syndrome (TTS).
- Report within 24 hours to the Vaccine Adverse Event Reporting System:https://vaers.hhs.gov/reportevent.html
- Alameda County providers should also email a copy of the VAERS report and the VAERS report number to COVIDreport@acgov.org.
Heparin and platelet transfusions should be avoided and alternative anticoagulant treatments should be used along with IVIG. Based on studies from Europe on people developing clots after the AstraZeneca vaccine, which is also an adenovirus-vector vaccine, the leading theory of pathogenesis is related to the development of platelet-activating autoantibodies against the platelet-factor 4 (PF4) protein.
Additional clinical guidance:
- CDC: Pathogenesis and Management of Thrombosis with Thrombocytopenia Syndrome (TTS) released April 23.
- UCSF COVID-19 Grand Rounds (recorded April 22): Dr. Andrew Leavitt discusses how to recognize and treat vaccine-induced thrombotic thrombocytopenia (about 43 minutes in, after a great talk with Dr. John Moore about the vaccine).
- American Society of Hematology (ASH) guidance for diagnosis and clinical management released April 16.
- CDC clinical health alert released April 13.
- ACIP slide deck from April 14.
- Sign up for Alameda County health alerts here.